|
This is always positive, since the degree of disorder increases in the transition from a liquid in a relatively small volume to a vapor or gas occupying a much larger space. The entropies of vaporization of a number of liquids to the same vapor volume are compared and show evidence that more or less ordered arrangements in. In thermodynamics, the entropy of vaporization is the increase in entropy upon vaporization of a liquid. 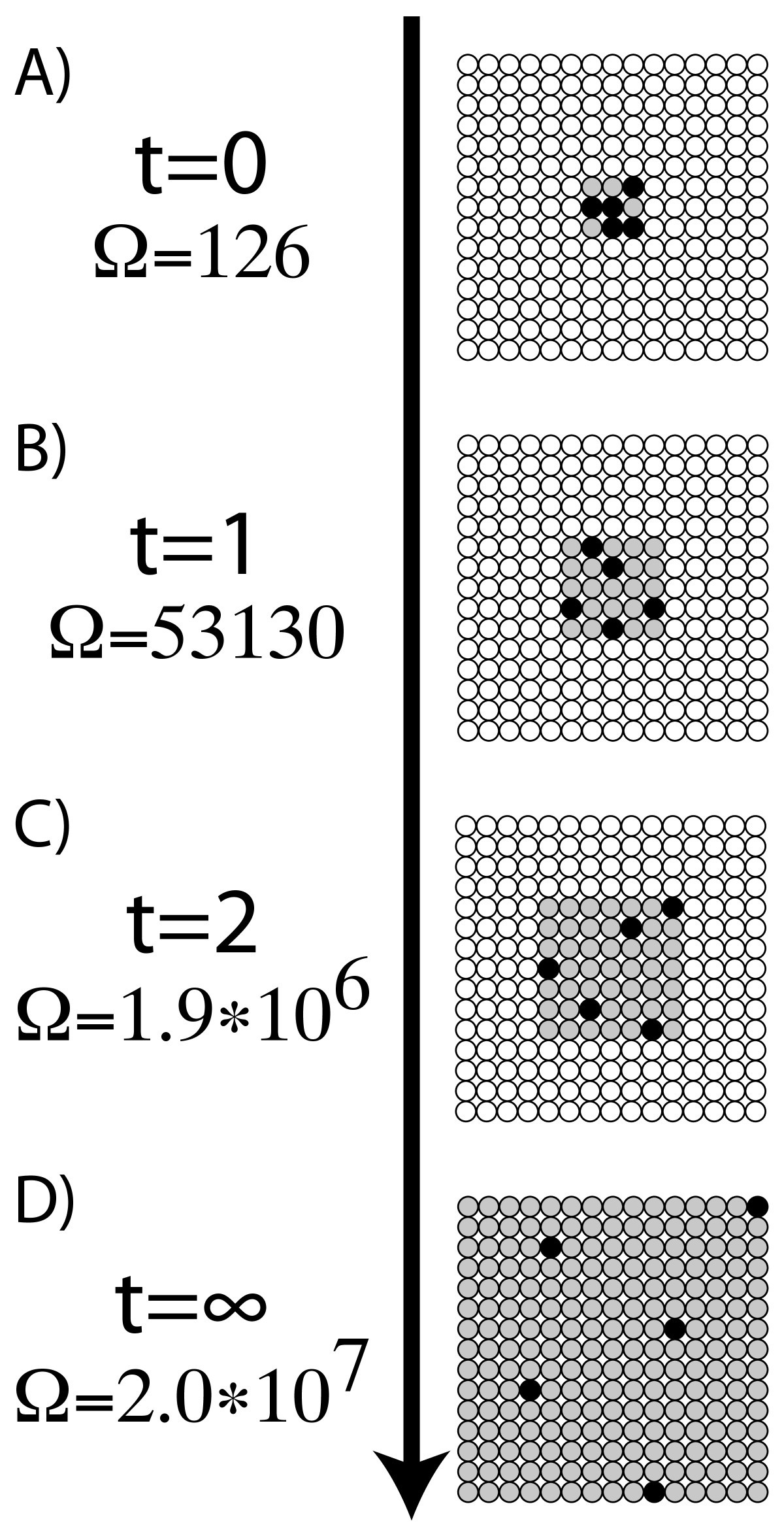
Δ G v a p = Δ H v a p − T v a p × Δ S v a p = 0 Īccording to Trouton's rule, the entropy of vaporization (at standard pressure) of most liquids is about 85 to 88 J mol -1 K -1.Not to be confused with Enthalpy of vaporization. 
In fact, you can convince yourself that using the enthalpy of vaporization of 40.68 kJ/mol from the reference as well as 373.15. As a reference, the actual value is around 109.02 J/K. Therefore, we have: Svap 40.7 kJ mol × 1 mol × 1000 J 1 kJ 373 K. Solve the equation (specific heat at constant pressure Cp 4.1818 kJ/Kkg). What is the boiling point(C) of a liquid with entropy of vaporization 68.3 JK1mol1 and enthalpy of vaporization 35.65 kJ mol1: rounded up to two decimal. Entropy of Vaporization using Troutons Rule calculator uses Entropy (4.5R)+(Rln(Temperature)) to calculate the Entropy, The Entropy of Vaporization. Since we must use T in K, T > 0 and everything is positive in this equation. Define final and initial temperature: Tf 20 ☌, Ti 100 ☌. g : denotes a saturated vapor state: l : denotes a saturated liquid state: The reference state for all property values is the liquid at the triple point, for which state the specific internal energy and the specific entropy have been set to zero. We will use the change in entropy formula: s Cp × ln (Tf / Ti), where Tf and Ti indicate the final and the initial temperature, respectively. In a phase transition such as vaporization, both phases coexist in equilibrium, so the difference in Gibbs free energy is equal to zero. specific entropy of vaporization : kJ/(kg K) Subscripts. Click here to get an answer to your question Calculate the boiling point of the liquid if its entropy of vaporization is 110JK-1mol-1 and the. 
At standard pressure P o = 1 bar, the value is denoted as Δ S o vap and normally expressed in J mol -1 K -1. This is always positive since the degree of disorder increases in the transition from a liquid in a relatively small volume to a vapor or gas occupying a much larger space. 
The entropy of vaporization is the increase in entropy upon vaporization of a liquid. If a substance has a molar heat of vaporization of 3.05 × 10 4 J / m o l and a normal boiling temperature of 80. Template:Distinguish Template:Unreferenced stub Enthalpy of vaporization refers to the physical destruction of an object that is exposed to intense heat. Measurements of the heat capacity of a substance and the enthalpies of fusion or vaporization can be used to calculate the changes in entropy that accompany a physical change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
